 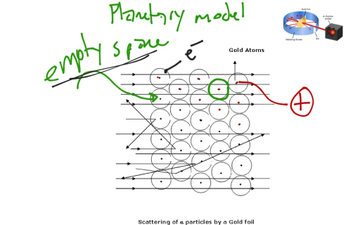
(a) Homo atomic Molecules – If a molecule has only one type of atom, then it is called homo atomic Molecules.Įxample : N 2 (Nitrogen Molecule), O 2 (oxygen Molecule), Cl 2 (Chlorine Molecule) etc. That is the smallest particle of an element or compound, which has an independent existence possible in nature, is called an molecules. Molecule – Two or more atoms that form molecules by a certain ratio. An atom is the smallest particle of a substance that participates in its chemical reaction, it is called an atom.Įxample : H (Hydrogen), Fe (Iron), Ca (Calcium), O (oxygen) etc.Ģ. Atom – The atom is the basic unit that makes up matter. The ratio of the number of atoms of its constituent elements in a compound is fixed.Īfter Dalton, in 1811, an Italian chemist Amedeo Avogadro explained the difference between the atom and the molecule, saying that there are two types of microscopic particles of any substance.ġ. A compound is formed by the addition of an atom of two or more elements.Chemical reactions are a rearrangement of atoms.The relative numbers and types of any compound atoms are fixed.The atoms of different elements are compounded by adding them in proportion to the smallest whole number. Atoms of the same element are similar to each other in size, mass and all other properties, but atoms of different elements are different.The atom is inseparable, meaning the atom cannot be divided into any more subtle particles, Which are neither produced nor destroyed in chemical reactions.An element is made up of many microscopic particles, it is called an atom.Following are the main points of this theory – John Dalton presented this theory in 1808 based on his experiments. Dalton’s atomic theory, Rutherford atomic model, quantum mechanical model, Bohr’s model Dalton’s Atomic Theory In this chapter we learn that what makes the atom of one element different from the atom of another element? Are atoms really indivisible, as proposed by Dalton, or are there smaller constituents inside the atom? We also read about Dalton’s atomic theory, Rutherford model, Bohr’s model and quantum mechanical model. Atomic Theory | Dalton’s, Rutherford Atomic Model, Bohr’s model, Quantum Mechanical Model We knows that atoms and molecules are the fundamental building blocks of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
